
This causes a deviation from ideal geometry (an H–C–H bond angle of 116. 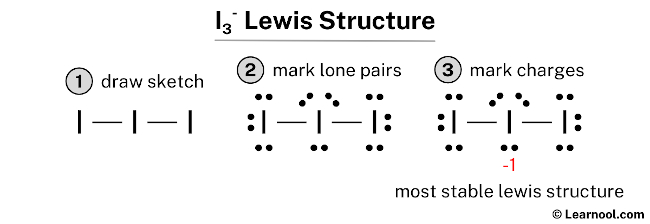
For example, in a molecule such as CH 2O (AX 3), whose structure is shown below, the double bond repels the single bonds more strongly than the single bonds repel each other. Determine the Lewis structure, molecular geometry and polarity for molecules using Lewis structures. This is because a multiple bond has a higher electron density than a single bond, so its electrons occupy more space than those of a single bond. Like lone pairs of electrons, multiple bonds occupy more space around the central atom than a single bond, which can cause other bond angles to be somewhat smaller than expected. However, if you have to describe the ion, you can use the phrase the like a polar molecule because I3- is soluble in water. (CC BY-NC-SA anonymous)Īs with SO 2, this composite model of electron distribution and negative electrostatic potential in ammonia shows that a lone pair of electrons occupies a larger region of space around the nitrogen atom than does a bonding pair of electrons that is shared with a hydrogen atom. An explanation of the molecular geometry for the I3 - ion (Triiodide Ion) including a description of the I3 - bond angles. The bond angles formed between each atom are close to 90 degrees. But the molecular shape of IF3 is T-shaped. There are six electron groups around the central atom, each a bonding pair. With an expanded valence, that this species is an exception to the octet rule. It gives information about the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. The electron geometry of IF3 is trigonal bipyramidal. The central atom, sulfur, contributes six valence electrons, and each fluorine atom has seven valence electrons, so the Lewis electron structure is. Molecular geometry is the three-dimensional arrangement of the atoms that constitute a molecule. \): The Difference in the Space Occupied by a Lone Pair of Electrons and by a Bonding Pair. The molecular shape does not take into account the lone pairs while molecular geometry does take lone pairs when determining the geometry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
